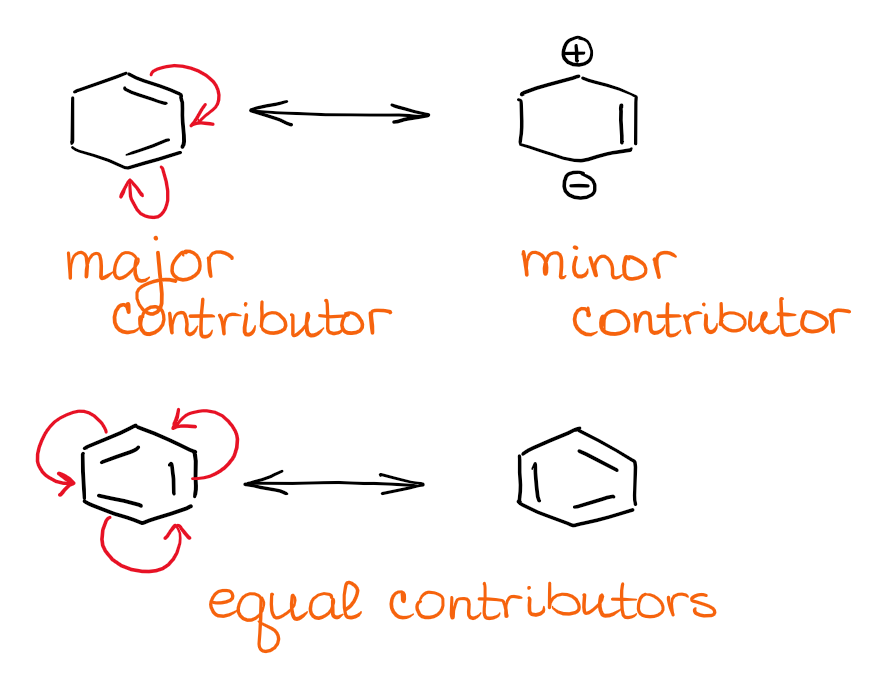
The structure with the least separation of formal charge is more stable. 
The structure with the least number of formal charges is more stable.The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets.Rules for estimating stability of resonance structures Are they equal or is there one which is more dominant? 
Now my problem is estimating the dominance of each form. Indicate the electron shift by a curved arrow, as shown in the image below.If I’m correct, pyracyclene has four resonance forms (double carbon bonds shown in red): Thus, the negative charge eventually transfers from one oxygen atom (O3) to another (O2). Since it acquires a lone pair, O2 will have a negative charge. This pi bond will break, and the electrons will transfer to the oxygen atom. However, there is another pi bond between nitrogen and oxygen (O2). 
The donor atom becomes neutral and forms a new pi bond with nitrogen, which now has 10 electrons in its valence shell. Step 2: Convert one lone pair from a charged oxygen (O3) atom to a bond with nitrogen. The nitrogen and all oxygen atoms obey the octet rule. The nitrogen has no lone pairs and has a charge +1. Another oxygen atom (O2) has two lone pairs, is double bonded to nitrogen, and has no charge. Also, these two oxygen atoms have a -1 charge and are single bonded to the nitrogen atom. The image shows that NO 3 – has two oxygen atoms (O1 and O3) with three lone pairs. The procedure is discussed in our Lewis Structure article. Step 1: Draw the Lewis dot structure of NO 3 –, as shown below. Resonance Structures of Nitrate Ion (NO 3 –) Structures with complete octet are more significant than incomplete ones.īased on the above rules, let us try to draw the resonance structures of the nitrate ion (NO 3 –).Neutral structures are more significant than charged structures. The structure with the least number of formal charges is more stable than those with more.Formal charges of each atom can identify the most significant and contributing resonance structure.The atoms remain fixed in their positions. Only electrons move through the molecules.There is no change in hybridization between the structures.They must be valid Lewis dot structures.They must have the same number of electrons.In order to draw the resonance structures, one has to keep the following rules in mind: Resonance Structures Examples How to Draw Resonance Structure Resonance structures occur in a molecule with multiple ways to place the lone pairs and pi bonds. These structures are collectively known as resonance structures. As a result of resonance, the molecule can have more than one structure that differs in the delocalized electrons’ position and bond formation. It is a phenomenon that explains the shifting of non-bonding electrons and pi bonds within the molecule. Resonance is a technique of describing the delocalized electrons in a molecule or ion that a single Lewis structure cannot describe. Sometimes more than one Lewis structure is needed to represent the molecular structure. However, a single Lewis structure cannot explain chemical bonding due to partial charges on the atoms and fractional bond order. We have learned that Lewis structure is a straightforward representation of valence shell electrons in an atom, ion, or molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
